天津工生所等在大肠杆菌合成藏红花素方面取得进展
藏红花酸和藏红花素是藏红花中的主要活性成分,具有抗肿瘤、抗氧化、抗高血压、抗动脉粥样硬化和抗抑郁等多种药理作用,在医药、食品、保健、印染等领域具有广泛应用。
中国科学院天津工业生物技术研究所研究员毕昌昊、孙媛霞、张学礼带领的研究团队和华东理工大学进行合作,在大肠杆菌合成藏红花素方面取得进展。该研究首先在产玉米黄素的重组大肠杆菌中引入不同裂解双加氧酶CCD和醛脱氢酶ALD,构建了新的藏红花酸合成途径,同时运用RBS文库调控和CRISPR-Cas9技术进行调控表达优化,并通过建立合适的分离检测方法,筛选获得藏红花酸产量高达4.42mg/L(0.51mg/g DCW)的高产菌株。随后,研究人员将糖基转移酶UGTs代谢途径整合到藏红花酸大肠杆菌生产菌株中,成功获得藏红花素(crocin-5)细胞工厂。
该研究利用大肠杆菌构建了高效合成藏红花酸和藏红花素的细胞工厂,为这两种产品的规模生产奠定了基础。
该研究获得中科院重点部署项目、国家自然科学基金和中央高校基本科研基金资助。相关研究成果发表在Microbial Cell Factories杂志上。
下面是论文原文的摘要、介绍和结论:
Construction of Escherichia coli cell factories for crocin biosynthesis
Wen Wang, Ping He, Dongdong Zhao, Lijun Ye, Longhai Dai, Xueli Zhang, Yuanxia Sun, Jing Zheng & Changhao Bi
Microbial Cell Factoriesvolume 18, Article number: 120 (2019)
Abstract
Background
Crocin is a carotenoid-derived natural product found in the stigma of Crocus spp., which has great potential in medicine, food and cosmetics. In recent years, microbial production of crocin has drawn increasing attention, but there were no reports of successful implementation. Escherichia coli has been engineered to produce various carotenoids, including lycopene, β-carotene and astaxanthin. Therefore, we intended to construct E. coli cell factories for crocin biosynthesis.
Results
In this study, a heterologous crocetin and crocin synthesis pathway was first constructed in E. coli. Firstly, the three different zeaxanthin-cleaving dioxygenases CsZCD, CsCCD2 from Crocus sativus, and CaCCD2 from Crocus ancyrensis, as well as the glycosyltransferases UGT94E5 and UGT75L6 from Gardenia jasminoides, were introduced into zeaxanthin-producing E. coli cells. The results showed that CsCCD2 catalyzed the synthesis of crocetin dialdehyde. Next, the aldehyde dehydrogenases ALD3, ALD6 and ALD9 from Crocus sativus and ALD8 from Neurospora crassa were tested for crocetin dialdehyde oxidation, and we were able to produce 4.42 mg/L crocetin using strain YL4(pCsCCD2-UGT94E5-UGT75L6,pTrc-ALD8). Glycosyltransferases from diverse sources were screened by in vitro enzyme activity assays. The results showed that crocin and its various derivatives could be obtained using the glycosyltransferases YjiC, YdhE and YojK from Bacillus subtilis, and the corresponding genes were introduced into the previously constructed crocetin-producing strain. Finally, crocin-5 was detected among the fermentation products of strain YL4(pCsCCD2-UGT94E5-UGT75L6,pTrc-ALD8,pET28a-YjiC-YdhE-YojK) using HPLC and LC–ESI–MS.
Conclusions
A heterologous crocin synthesis pathway was constructed in vitro, using glycosyltransferases from the Bacillus subtilis instead of the original plant glycosyltransferases, and a crocetin and crocin-5 producing E. coli cell factory was obtained. This research provides a foundation for the large-scale production of crocetin and crocin in E. coli cell factories.
Introduction
Crocin is the most valuable component of the Crocus sativus stigmas. It has an unsaturated conjugated polyenoic acid structure, derived from carotenoids [1]. Crocin has high medicinal value, with excellent anti-apoptotic [2,3,4], anti-hyperlipidemic [5], anti-atherosclerotic [6] and antioxidant effects [7, 8]. Additionally, crocin has been proved to have a significant inhibitory effect against a variety of cancer cells [9,10,11], and can reduce the side effects of antitumor drugs such as cisplatin and improve their efficacy [12]. Animal experiments have demonstrated that treatment of female rats suffering from colon adenocarcinoma with crocin isolated from saffron prolonged their survival and inhibited the growth of the tumors [13]. At present, crocin manufacture mainly depends on complicated extraction and purification from the saffron stigma. The resulting product has low purity, and the manufacturing process requires the large-scale cultivation of saffron crocus, which can destroy the natural environment. The lack of availability has been the main factor limiting the broader application of crocin. Therefore, it is desirable to develop more economical and environmentally friendly ways to produce crocin.
With the development of synthetic biology, biosynthesis of crocin from simple carbon sources in fermentation facilities with engineered microbes heterologously expressing genes or enzymes of interest has become a promising complement to traditional sources. At present, the biosynthetic pathway of crocin has been partially elucidated [14], and the technical difficulties lie in the screening of some key enzyme genes and regulating the synthesis of the corresponding proteins. The proposed crocin biosynthesis pathway (Fig. 1) starting from zeaxanthin contains three major steps, catalyzed by a carotenoid-cleaving dioxygenase (CsCCD2), an aldehyde dehydrogenase (ALD8) and UDP-glucuronosyltransferases (YjiC, YdhE, YojK), respectively [15,16,17]. Nevertheless, the glycosylation steps might require a variety of UGTs to obtain multiple structural forms (crocin-1 through -5) by sequential glycosylation. These forms encompass the crocetin mono-β-glucosyl ester (crocin-5), the crocetin β-glucosyl-β-gentiobiosyl ester (crocin-2), the crocetin di-β-glucosyl ester (crocin-3) and the crocetin mono-β-gentiobiosyl ester (crocin-4).
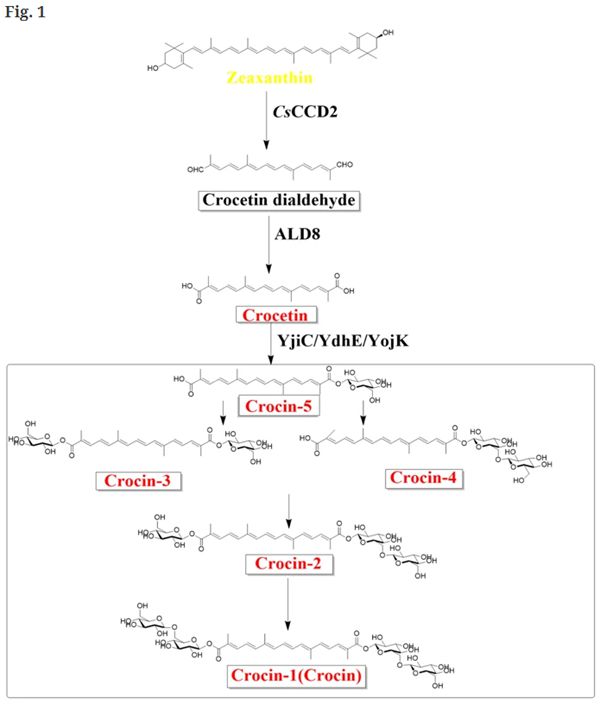
Fig. 1 Heterologous crocetin and crocin biosynthesis pathways initiated from zeaxanthin. Crocin-1, crocin-2, crocin-3 or crocin-4 and crocin-5 were obtained in vitro, and only crocin-5 was obtained in E. coli cells
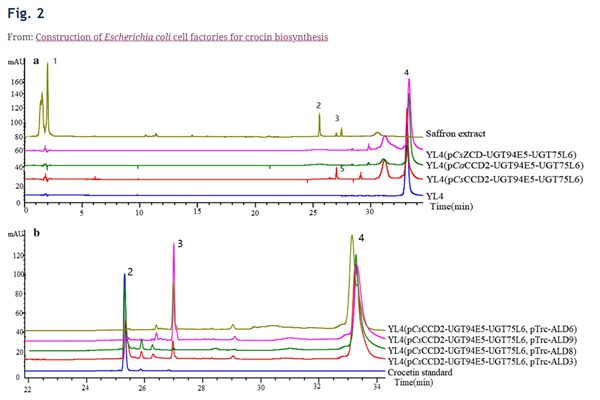
Fig. 2 The analysis of fermentation products of the engineered strains by HPLC. peak 1: Crocin (tR = 2.00 min); peak 2: Crocetin (tR = 25.32 min); peak 3 and 5: Crocetin dialdehyde (tR = 27.05 min); peak 4: Zeaxanthin (tR = 33.30 min); tR: Retention time. a From top to bottom were Saffron extract, YL4(pCsZCD-UGT94E5-UGT75L6), YL4(pCaCCD2-UGT94E5-UGT75L6), YL4(pCsCCD2-UGT94E5-UGT75L6) and YL4. b From top to bottom were YL4(pCsCCD2-UGT94E5-UGT75L6, pTrc-ALD6), YL4(pCsCCD2-UGT94E5-UGT75L6, pTrc-ALD9), YL4(pCsCCD2-UGT94E5-UGT75L6, pTrc-ALD8), YL4(pCsCCD2-UGT94E5-UGT75L6, pTrc-ALD3) and Crocetin standard
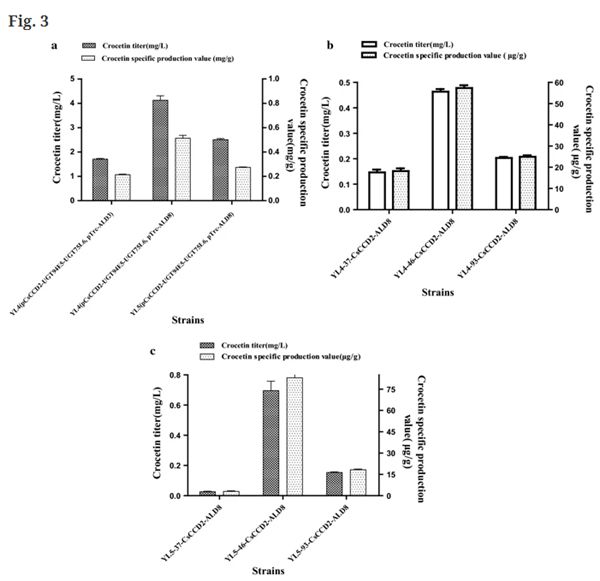
Fig. 3 Further improvement of crocetin production. a Effects of parent strains and ALDs on crocetin titers and specific production values. b Crocetin titers and specific production values with different promoters. c Crocetin yields obtained using strains with different zeaxanthin yields
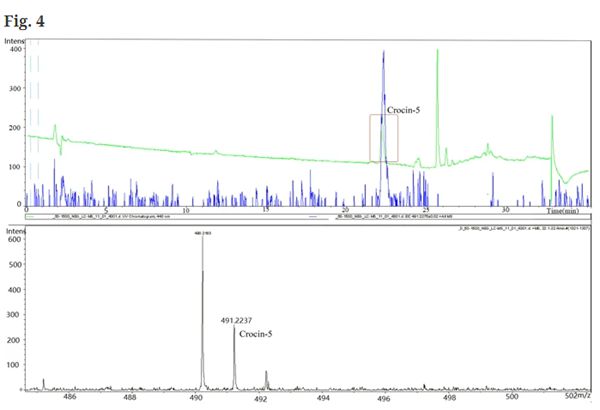
Fig. 4 The analysis of the fermentation products of strain. YL4(pCsCCD2UGT94E5UGT75L6,pTrcALD8,pET28a-YjiC-YdhE-YojK). Crocin-5 (tR = 22.10 min, m/z = 491.22) was detected in the extract after 72 h of cultivation by HPLC and LC–ESI–MS
Conclusions
In this study, a heterologous crocin synthesis pathway was constructed in vitro using purified glycosyltransferases from bacterium Bacillus subtilis rather than the original plant glycosyltransferases. Subsequently, a crocetin and crocin-5 producing E. coli cell factory was obtained for the first time. Firstly, three different zeaxanthin-cleaving dioxygenases, the genes CsZCD, CsCCD2 from Crocus sativus, and CaCCD2 from Crocus ancyrensis, as well as the glycosyltransferases UGT94E5 and UGT75L6 from Gardenia jasminoides, were respectively introduced into zeaxanthin-producing E. coli cells. The results showed that CsCCD2 catalyzed the synthesis of crocetin dialdehyde. Next, the aldehyde dehydrogenases ALD3, ALD6 and ALD9 from Crocus sativus, as well as ALD8 from Neurospora crassa were tested for crocetin dialdehyde oxidation. It was found that ALD3 and ALD8 could produce crocetin in E. coli, whereby the latter had higher activity. Besides, the medium and fermentation conditions were also analyzed and a lower fermentation temperature of around 20 °C was found to be optimal. Using the optimized conditions, we were able to produce 4.42 mg/L crocetin using the strain YL4(pCsCCD2-UGT94E5-UGT75L6,pTrc-ALD8), which was significantly higher than the previous report of an engineered yeast strain with a titer of 1.22 mg/L in shake-flask culture [20].
Since no crocin was obtained using the plant-derived glycosyltransferases UGT94E5 and UGT75L6, similar enzymes from diverse sources were screened in vitro activity assays. The results showed that crocin and its various derivatives can be produced by the glycosyltransferases YjiC, YdhE and YojK from Bacillus subtilis. Subsequently, these genes were introduced into the previously constructed crocetin producing strain. Finally, crocin-5 was detected by HPLC and LC–ESI–MS in the products of strain YL4(pCsCCD2-UGT94E5-UGT75L6,pTrc-ALD8, pET28a-YjiC-YdhE-YojK).
Since crocin is glycosylated products derived from crocetin with four glucose groups, the derivatives of crocin are crocetin with different numbers of glucose groups in different patterns. Normally, since these derivatives have a same core, so that they may have different chemical properties and catabolic characteristics, but their medical effect should be similar. Although there was not medical reports concerning crocin-5, we speculated crocin-5 might have some if not all crocin medical activities. This research provides a foundation for the large-scale production of crocetin and crocin using E. coli cell factories.
相关内容
标签:科研 藏红花素
声明:
1、藏红花网登载此文出于传递更多信息之目的,并不意味着赞同其观点或证实其描述。医学相关、具体治疗及选购请咨询医生或相关专业人士。
2、本站部分图片整理自网络,无法核实真实出处,仅为分享知识,如涉及侵权,请第一时间联系小编删除,谢谢!






